977
Views & Citations10
Likes & Shares
Mother’s milk is known for centuries to be beneficial for infants and to
prevent many diseases, including malaria. What are the constituents of breast
milk responsible for this efficacy? Do formula milk, cow milk or soy milk has same properties?
Keywords: Mother’s milk,
Formula milk,
Infants,
Diseases, Cow
milk, Soy
milk
A FEW HISTORICAL DATA
It all begins in 1952 with
the work of the Liverpool School of Tropical Medicine [1,2].
They found that in rats
inoculated with Plasmodium berghei and
living on a diet of milk there was a strong suppression of the growth of the
parasites. This was valid for whole cow’s milk, reconstituted dried milk from
different origins and human milk. Most rats on normal laboratory diet died in a
few days. The authors suggested that the milk might contain an inhibitory
substance and that herein lies the explanation of the common observation that
severe malaria is not often seen in very young infants. In these first
experiments only blood-transmitted malaria had been studied.
The London School of
Tropical Medicine followed-up on this early work and confirmed that a milk diet
had a suppressive action on Plasmodium
cynomogi in monkeys. But after a return to normal diet a severe
recrudescence took place [3].
In a more recent study, 137
infants exclusively breast fed and 358 control infants from the Democratic
Republic of the Congo were assessed for fever and malaria infections by
polymerase chain reaction, at 6 months of age. Breast feeding was significantly
associated with a reduced risk of clinical malaria [4].
The World Health
Organization now recommends exclusive breastfeeding for the first 6 months of
life.
NOMADS AND TROPICAL DISEASES
Fulani are a widely spread
African ethnic group characterized by lower susceptibility to Plasmodium falciparum and clinical
malaria morbidity. They are characterized by a higher rate of lactase
persistance. This trait is common in Europe and certain African people with
traditions of raising cattle. Lactase non-persistance in other African tribes
is often called lactase intolerance. The potential immunoprotective properties
of dietary cow milk as a reason for the malaria resistance of Fulani warrant
further investigation. [5]
Milk-drinking African nomads
show an unusual freedom from infection with Entamoeba
histolytica compared with similar nomads taking a mixed diet. The authors
related this to a low content in iron in cow’s milk. A personal communication
from Dr. Patrick Ogwang informs that in Uganda malaria is highest in East and
North Uganda where the staple food is cereals with high iron content, in
western Uganda where milk and low iron foods are eaten most malaria is low. In
the past however, people in East and North also kept cows (zebu) and took milk
regularly and malaria was not as rampant.
One of the first mistakes of
Western medicine in Africa was the iron supplementation to the Somali nomads in
1968. Blood analysis of these nomads had shown that according to European
standards they were suffering from anemia [6].
The incidence of infections was studied in 137 iron deficient Somali nomads, 67 of whom were treated with placebo and 71 with iron. Seven episodes of infection occurred in the placebo group and 36 in the group treated with iron; these 36 episodes included activation of pre-existing malaria, brucellosis and tuberculosis. This difference suggested that host defence against these infections was better during iron deficiency than during iron repletion.
In an outbreak of Plasmodium falciparum malaria following
re-feeding after famine cerebral malaria was restricted to children eating
grain. Those given grain were more likely to experience cerebral malaria. Nomad
children consuming a predominantly milk diet were free of this complication
despite an equivalent incidence of uncomplicated malaria. Freedom of nomads
from cerebral complications may be due to inhibition by the milk diet of rapid
division of the parasite combined with delayed recovery after famine of T cell
function [7].
Some early results
Formula-fed babies contract
gastroenteritis more than breast-fed babies, which is of concern to mothers who
cannot breastfeed or, as with HIV-infected mothers, are discouraged from
breastfeeding. The ability of endogenous breast milk xanthine oxidase to
generate the antimicrobial radical nitric oxide has been measured and its
influence on the growth of Escherichia
coli and Salmonella enteritides
examined. Breast milk, but not formula feed, generated nitric oxide. Xanthine
oxidase activity substantially inhibited the growth of both bacteria. An
important natural antibiotic system is missing in formula feeds; the addition
of xanthine oxidase may improve formula for use when breastfeeding is not a
safe option [8].
Work done on the shores of Lake
Victoria suggested that milk might be deficient in certain nutrients required
by the parasite, but that these are present in any normal diet. For the first
three months in life children are exclusively breast-fed and they stay malaria
free. At the third or fourth month mothers usually start giving them a variety
of foods in addition to the breast milk and these extras might supply the
nutrients required by the parasite.
In 1983 a five month study was
undertaken in Brazil to test the effect of a total milk diet on the
susceptibility of mice to various doses of a the rodent malaria P. berghei. The development of humoral
immunity was followed by quantitation of the specific serum immunoglobulins
(IgG and IgM). High levels of IgG antibodies persisted for 150 days, IgM
antibodies were only observed during the two first weeks of infection. The
results indicated that a milk diet administered to mice as the only source of
food protected them against fatal malaria infection regardless of the number of
parasites inoculated. The acquired immunity was still present in the mice at
150 days post inoculation. [9,10].
But these very promising
treatments were ignored by BigPharma and subsidized research. Obviously dairy
milk or dried milk is not an interesting cash cow in the fight against malaria.
The detrimental role of para-aminobenzoic acid (PABA)
In the light of resistance to
most, if not all, of the pharmaceutical molecules (chloroquine, amodiaquine,
lumefantrine, chloroquine, sulfadoxine-pyrimethmine) some research groups have
tried to better understand all these fifty years old findings. And the proposed
detrimental role of PABA (p-aminobenzoic acid) seems to be confirmed. PABA is a
precursor of folic acid.
A large scale clinical trial was
run on 25 000 infants in Pemba, Zanzibar. The iron and folic acid-containing
groups of the trial had to be stopped on the recommendation of the data and
safety monitoring board. It resulted in an increased risk of severe illness and
death [11-15].
The National Institute for
Medical Research finds that milk contains very little p-aminobenzoic acid
(PABA), not more than 0.004 ppm [16].
This substance is much more
plentiful in some of the constituents of a good laboratory diet, where the
vegetal components contain up to 60 ppm of PABA. In vitro Plasmodium requires
PABA for satisfactory growth. Experiments were undertaken to study this
possibility. Rats were maintained on different diets: normal diet, milk, milk
with 1000 ppm of PABA. Only the rats on the milk diet stayed free of Plasmodium berghei on day 12. Plasmodium
requires exogenous dietary PABA for survival [17].
Plasmodium generates its own
PABA in its apicomplexan organelle, but not in quantities sufficient to
guarantee survival and multiplication of trophozoites and schizonts in the
infected erythrocyte. And as the human body does not generate its own PABA or
folates, but takes them essentially from green vegetables, the parasite has to
rely on this supply. If the diet is exclusively on milk it has no chance to
survive.
PABA is an intermediate in the
synthesis of folate. And the folate supplements sold in our nutrition might be
harmful to African new-borns infected by malaria [18].
Iron, zinc, potassium, selenium
Iron is essential for the
survival and multiplication of the Plasmodium parasite. In humans iron
deficiency appears to protect against severe malaria while iron supplementation
increases risks of infection and disease.
Anemia may even protect against
malaria as it was found at the University of North Carolina. Researchers
studied the red blood cells of 135 anemic children aged 6-24 months in a
malaria-endemic region of The Gambia and confirmed that anemia offers greater
natural protection against blood-stage malaria infection than sickle-cell trait
[19].
It would thus be advisable to
keep the iron concentrations rather on the low side. This can be achieved by
drinking milk. Lactoferrin, a glycoprotein found in milk, has the ability to
bind Fe ions with high affinity and to regulate iron distribution within the body
[20].
Zinc is an essential element
with strong bactericidal properties and very efficient against diarrhea and
other diseases. UNICEF estimates that a formula-fed child living in unhygienic
conditions is between 6 and 25 times more likely to die of diarrhea and four
times more likely to die of pneumonia than a breastfed child. Zinc stimulates
the immune system and increases CD4. Most medical plants like Artemisia are
rich in zinc [21].
This might be one of the reasons
why human milk is rich in zinc. The zinc content of milk varies with species,
lower in cow milk, and stage of lactation, much higher in colostrum. This is
probably contributing to the immunity of new-borns against malaria. Variations
in zinc absorption from different milks and formulas employed in infant feeding
are of serious concern [22].
There is considerable evidence
to suggest that the bioavailability of zinc from human milk is especially
favorable. It has been confirmed with radioactive zinc studies in adults in
whom absorption with mature human milk averaged 57% compared with 32% for cow’s
milk. Hence, zinc plasma concentrations of infants fed with cow’s milk-based
infant formula was significantly lower. Zinc absorption from soy-based infant
formulas is especially poor. The poor absorption of zinc from soy formulas has
been found to be attributable to the phytate present in these formulas.
Potassium concentrations in
mother’s milk are 2 times higher at postpartum in colostrum than one month
later in mature milk. It is likely that potassium plays a key role. The
potassium concentration in the plasma of neonates is much higher than in the
plasma of the mothers: 5.9 mmol/l versus 3.8 mmol/l [23,24].
The content of selenium in
colostrum is significantly higher (28.6 ng/ml) than that in mature milk (15.1
ng/ml) [25].
Fats, taurine, linoleic acid, oxidants
Mother’s milk is rich in fats:
4.4 % versus 3.3% in bovine milk. A fatty diet kills the sporozoites in the
hepatocytes by mediating oxidative stress [26].
And rich in linoleic acid, a
strong antimalarial. Linoleic may also act as a growth promotor in the neonate.
Concentration was quantified in human milk and infant formula. Concentration of
the biologically important conjugated linoleic acid in human milk ranged from
2.23 to 5.43 mg/g; that of formula from undetectable to 2.04 mg/g fat [27,28].
Arachidonic acid and
docosahexaeonic acid, n-3 and n-6 long chain polyunsaturated fatty acids, are
well present in mother’s milk but are absent from many infant formulas. During
neonatal life, there is a rapid accretion of arachidonic and docosahexaenoic
acid in infant brain. Cognitive development of breast-fed infants is generally
better [29-32].
Arachidonic acid also has strong
antimalarial properties via PGE production. Already in 2000 it had been demonstrated
in a study on Gabonese children with and without malaria that prostaglandins
are important pro-inflammatory mediators of the host-immune response to
infection [33].
The concentration of arachidonic
acid is on the average 0.5% by wt in breast milk. The IUPAC Lipid Handbook
confirms that human milk contains arachidonic acid, but cow's milk does not
[34-37].
Arachidonic acid is much higher
in breast milk than in formula milk or bovine milk [38,39].
Immunoglobulins
Mother’s milk is rich in
taurine: 358 mg/kg. Cow’s milk only contains 50 mg/kg, formula milk 30. Taurine
has a strong effect on immunity. Replacement 50% of the sulfated amino acid
methionine from plant origin by taurine from mammal origin doubles IgA in
broilers and increases IgM by 50% [40-42].
Neonates and young infants up to
6 months are relatively protected against symptomatic malaria. The prevailing
paradigm was that maternal antimalarial antibodies transferred to the fetus in
the last trimester of pregnancy protect the infant from early infections.
However direct evidence and research results do not support this paradigm [43].
The mystery of the invasion of
hepatocytes through Kupffer cells may eventually find an answer in this
context. Kupffer cells are specialized macrophages and protect the liver
against microbes, contaminants and other aggressions. Why these phagocytes are
used as entry gate by sporozoites indeed is difficult to understand [44].
Some studies have shown that IgA
antibodies preferentially attach to hepatocytes, blocking the entry for
sporozoites. Their number on the surface of Kupffer cells is much lower, 10%
versus 63% on hepatocytes. If so, it is logical to expect that taurine has
prophylactic antimalarial properties [45].
Breast milk is a remarkably
“altruistic” secretion, that is, its contents are directed at protecting the
infant with minimal benefit to the mother. The concentration of antibodies,
mainly IgA, is 10-100 folds higher than in serum. In colostrum it is as high as
90 g/L.
Mother’s milk is rich in
hydrogen peroxide in the first postpartum week. Hydrogen peroxide like other
ROS kills parasites [46].
Estimation of nitrate and
nitrite concentrations of milk sources may provide another insight. In
colostrum (1-3 days postpartum) nitrite concentrations are much higher than in
mature milk (0.08 mg/100 mL versus 0.001) [47].
According to the authors this
change is partly due to the changing intestinal microflora in the baby and the
changing metabolic demands as the baby grows. The beneficial effects of NO in
adult stomachs on gastroprotective and immunomodulatory functions are known.
Arginine plays a key role in the metabolism of nitrates. Therefore, it is
reasonable to surmise that nitrite must be supplied to the newborn by
colostrum. A recent thesis from Sweden confirms and documents well all these
positive elements. Dietary nitrates have potent anti-inflammatory effects,
without impairing the ability to clear an infection. They are able to restore
the gastric and colonic mucus layer in case of colitis [48].
In breastfed infants “good
bacteria” of the gut are important in determining the “direction” of maturation
of immunity. Together with other maternal and infant factors, the breastfed
infant's mucosal and systemic immune responses are influenced by a different micro
eco milieu of the gut compared to the formula fed infant. An environment that
does not encourage the hatching of Trichuris
trichuria eggs due to the absence of the required ‘pro hatching’ bacteria. Escherichia coli in the gut in the
breastfed, is deemed another indirect anti-parasitic potential that lies within
breast milk [49].
Several protector mechanisms
have been proposed for Lactobacillus against gastroenteritis. The most likely
mechanism is its role as immunomodulator. Higher bottle feeding with milk
poorer than breast milk in Lactobacillus increases the risk of diarrhea. In a
trial probiotics Lactobacillus and Bifidobacterium shortened duration of
diarrhea to 34.1 h versus 58 h with placebo and reduced the number of stools
(7.3 vs. 15.9 with placebo) [50-53].
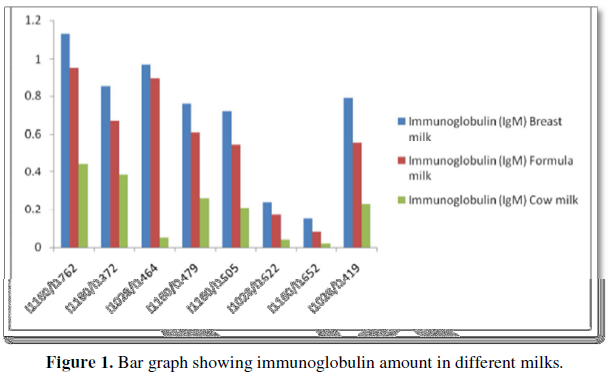
Immunoglobulins are much higher in breast milk than in formula or cow milk (Figure 1).
Lactoferrin, the best vaccine against malaria
Breast milk also
contains lactoferrin, 5-13 g/L in colostrum and 2-4 g/L in milk. The
concentration of lactoferrin in milk varies widely from one species to another.
It is highest in human milk (2 g/L, 25x MI, moderate in murine milk (0.28 mg/L,
3.5x M) and very low in ruminant milk (-0.01 mg/L in bovine milk, 0.12 × 10-6
M). There is an international trend toward the addition of lactoferrin to
infant formula [54,55].
The bactericidal and
bacteriostatic properties of lactoferrin are well known Lactoferrin indeed
binds strongly to iron, and almost irreversibly. This complex in a dose
dependent manner enhances ROS production. A variety of free radical ions
inhibits a variety of tumors, intracellular parasites and microbes.
A lower expression of
the multidrug resistant gene (MDR1) is noticed. This can be a helpful in
decreasing the resistance mechanisms of pathogens.
In parallel there is
a 4-fold increase in phagocytic capacity of macrophages. Mouse peritoneal
macrophages or human blood monocytes co-cultured with intracellular forms of Trypanosoma cruzi in the presence of
human lactoferrin took up greater numbers of organisms than in the absence of
lactoferrin [56,57].
The binding of
lactoferrin to iron is 250 times higher than for the parent molecule
transferrin and down to a much lower pH. Iron is one on the most important
promoters of Plasmodium development [58].
Lactoferrin is to a
large extent destroyed at temperatures >60°C. Breast milk is thus by far
preferable to sterilized cow milk or powdered milk [59].
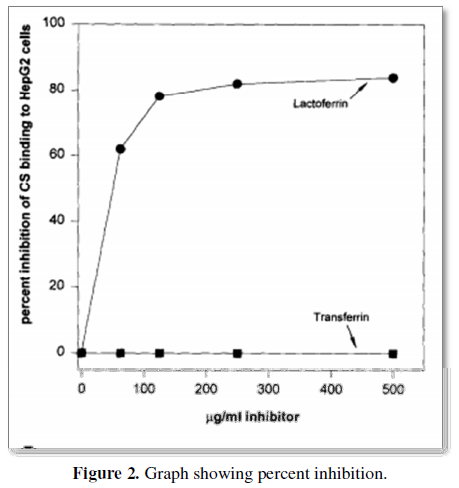
Very low density lipoproteins (VLDL), similarly to malaria sporozoites are removed from the blood circulation by the liver within minutes after injection by Anopheles mosquitoes. The sporozoite's surface is covered by the circumsporozoite protein (CS). Lactoferrin, a protein with antibacterial properties found in breast milk is also rapidly cleared from the circulation by hepatocytes in case of malaria infection. CS, lactoferrin and remnant VLDLs compete in vitro and in vivo for binding sites on liver cells [60] (Figure 2).
Other authors also
found that lactoferrin inhibits sporozoite invasion liver cells in a
concentration-dependent fashion. Up to 80%, this is much higher than the 32% of
the GSK-Bill Gates vaccine.
Studies have shown
that lactoferrin is the special constituent that allows iron-catalyzed toxic O₂
species to efficiently work their damage. Adding 10-8 M pure
lactoferrin has a significant impact on hemolysis. Lactoferrin seems
particularly suited to focus its products directly onto membranes of target
cells. Lactoferrin is highly cationic, which suggests that it might be readily
absorbable to negatively charged cellular membranes of erythrocytes.
Plasmodium falciparum extensively remodels its host red blood cell. The zeta potential is an
electrochemical property of cell surfaces that is determined by the net
electrical charge of molecules exposed at the surface of cell membranes. The
RBC membrane is negatively charged and is surrounded by a fixed layer of
cations. Using an electrophoretic mobility assay, it was found that the main
zeta potential was significantly lower in in RBCs infected with Plasmodium
falciparum [61,62].
Lactoferrin can also
be found in honey, generated by the metabolism of bees. Bee sting contains the
highest concentration of lactoferrin. It is secreted by the serous cells of the
major and minor salivary glands. It has an iron-chelating property which
deprives microorganisms of this essential element. In addition, lactoferrin has
demonstrated potent antiviral, antifungal and antiparasitic activity, towards a
broad spectrum of species. Lactoferrin exhibits in vitro anti-inflammatory
activities and several domains are present within its polypeptide chain that
demonstrates antimicrobial effects [63].
Mycobacterium
tuberculosis and most bacteria, except Borrelia (Lyme), must import iron from
its host for survival, and its siderophore-dependent iron acquisition pathways
are well established. Lactoferrin extracts excess iron from host proteins.
So maybe the message
of Melinda Gates in the Wall Street Journal of May 20, 2016 has a lot of merit “Many
newborn deaths can be prevented by simple, inexpensive measures, such as,
teaching women to breast-feed, which immediately gives a baby nutrients and
hydration, and guards against infection, one of the biggest killers of
newborns. Each year 2.9 million infants die in their first 28 days of life.”
But a press release
of April 28, 2017 shows that the Gates Foundation supports research into milk
to find new drug molecules which enhance the absorption and efficacy of
existing drugs. The interest of Bill Gates in milk is only to find new
molecules for the BigPharma business.
1. Maegraith
BG, Deegan T, Sherwood Jones E (1952) Suppression of malaria (P. berghei) by milk. Br Med J 2:
1382-1384.
2. Keppie
Audrey A (1953) Modified course of T.
congolense infection in mice given diets with milk casein. Br Med J 2:
853-857.
3. Bray
RS (1953) Effect of milk diet on P.
cynomolgi infections in monkeys. Br Med J 1: 1200-1201.
4. Brazeau
NF, Tabala M, Kiketa L (2016) Exclusive breastfeeding and clinical malaria risk
in 6 month old infants: A cross-sectional study from Kinshasa, Democratic
Republic of the Congo. Am J Trop Med Hyg 95: 827-830.
5. Lokki
I, Järvelä I, Holmberg V (2011) Lactase persistence genotypes and malaria
susceptibility in Fulani in Mali. Malaria J 10: 9.
6. Murray
MJ, Murray AB, Murray MB, Murray CJ (1978) The adverse effect of iron repletion
on the course of certain infections. Br Med J 2: 1113-1115.
7. Murray
MJ, Murray AB, Murray NJ, Murray MB (1978) Diet and cerebral malaria: The
effect of famine and re-feeding. Am J Clin Nutr 31: 57-61.
8. Stevens
CR, Millar TM, Clinch JG, Kanczler JM, Bodamyali T, et al. (2000) Antibacterial
properties of xanthine oxidase in human milk. Lancet 356: 829-830.
9. Ferraroni
JJ (1983) Efeito da dieta lactea na supressao da parasitemia. Mem Inst Oswaldo
Cruz 78: 27-35.
10. Murray
MJ, Murray A, Murray CJ (1980) The salutary effects of milk on amoebiasis and
its reversal by iron. Br Med J 280: 1351-1352.
11. Sazawal
S, Black RE, Ramsan M, Chwaya HM (2006) Effects of routine prophylactic supplementation
with iron and folic acid on admission to hospital and mortality in pre-school
children in a high malaria transmission setting: Community-based, randomised,
placebo-controlled trial. Lancet 367: 133-143.
12. McConkey
Glenn A (1999) Targeting the Shikimate pathway in the malaria parasite Plasmodium falciparum. Antimicrob Ag
Chemother 43: 175-177.
13. Kretschmar
W, Voller A (1973) Suppression of Plasmodium
falciparum malaria in Aotus monkeys by milk diet. Z Tropenmed Parasitol 24:
51-59.
14. Jacobs
RL (1964) Role of p-aminobenzoic acid in Plasmodium
berghei infection in the mouse. Exp Parasitol 15: 213-225.
15. Nowell
F (1970) The effect of a milk diet upon Plasmodium
berghei, Nuttallia (=Babesia) rodhaini and Trypanosoma brucei infections in mice. Parasitology 61: 425-433.
16. Hawking
F (1954) Milk p-aminobenzoate and malaria of rats and monkeys. Br Med J 1:
425-429.
17. Kicska
Gregory A, Ting LM, SchrammVern L, Kim K (2003) Effect of dietary
p-aminobenzoic acid on murine Plasmodium
yoelii infection. JID 188: 1776-1781.
18. Carter
JY, Loolpapit MP, Lema OE, Tome JL, Nagelkerke NJ, et al. (2005) Reduction of
the efficacy of antifolate antimalarial therapy by folic acid supplementation.
Am J Trop Med Hyg 73: 166-170.
19. Goheen
MM, Wegmüller R, Bah A, Darboe B, Danso E, et al. (2016) Anemia offers stronger
protection than sickle cell trait against the erythrocytic stage of Falciparum
malaria and this protection is reversed by iron supplementation. EBioMedicine
14: 123-130.
20. Baker
Heather M, Edward N (2004) Lactoferrin and iron: Structural and dynamic aspects
of binding and release. Biometals 17: 209-216.
21. Mocchegiani
E (2007) Zinc and ageing: Third Zincage conference Immunity & Ageing 4:
5.
22. Hambidge
K, Casey CE, Krebs NF (1986) In: Mertz W (ed) Trace elements in human and
animal nutrition. Academic Press: Orlando FL.
23. Silprasert
A, PPruenglampoo L (1991) Composition of sodium, potassium, calcium, magnesium
and phosphorus in human breast-milk at different stages of lactation period.
Available at: http://thaiagris.lib.ku.ac.th
24. Martinerie
L, Pussard E, Foix-L'Hélias L, Petit F, Cosson C, et al. (2009) Physiological
partial aldosterone resistance in human newborns. Pediatr Res 66: 323-328.
25. Kim
ES, Kim JS, Tamari Y (1998) Quantitation of taurine and selenium levels in
human milk. Adv Exp Med Biol 442: 477-486.
26. Zusarte-Luis
V, Mota MM (2017) Dietary alterations modulate susceptibility to Plasmodium
infection. Nat Microbiol Lett.
27. Mc
Guire MK, Park Y, Behre RA, Harrison LY, Shultz TD, et al. (1997) Conjugated
linoleic acid concentrations of human milk and infant formula. Nutr Res 17:
1277-1283.
28. Brenna
T, Varamini B, Jensen RG (2007) Docosahexaenoic and arachidonic acid
concentrations in human breast milk worldwide. Am J Clin Nutr 85: 1457-1464.
29. Fleith
M, Clandinin MT (2005) Dietary PUFA for preterm and term infants: Review of
clinical studies. Crit Rev Food Sci Nutr 45: 205-229.
30. Lauritzen
L, Carlson SE (2011) Maternal fatty acid status during pregnancy and lactation
and relation to newborn and infant status. Matern Child Nutr 2: 41-58.
31. Innis
SM (2007) Human milk: Maternal dietary lipids and infant development. Proc Nutr
Soc 66: 397-404.
32. Douglas
JP, Peter GK, Brice WJ (2001) Inverse relationship of plasma prostaglandin E2
and blood mononuclear cell cyclooxygenase-2 with disease severity in children
with Plasmodium falciparum malaria. J
Infect Dis 183: 113-118.
33. Brenna
JT, Varamini B, Jensen RG, Diersen-Schade DA, Arterburn LM (2007)
Docosahexaeonic and arachidonic acid concentrations in human breast milk
worldwide. Am J Clin Nutr 85: 1457-1464.
34. Kim
H, Kang S, Jung BM (2017) Brest milk fatty acid composition and fatty acid
intake of lactating mothers in South Korea. Br J Nutr 13: 1-6.
35. Henjum
S, Lie O, Chandyo RK, Kiellevold M (2017) Erythrocyte fatty acid composition of
Nepal breast-fed infants. Eur J Nutr 57:1003-1013.
36. Koletzko
B (2016) Human milk lipids. Ann Nutr Metab 69: 28-40.
37. Sueyoshi
Y, Oda H (1963) Comparative study on amounts of polyunsaturated fatty acids in
human and cow’s milk. Keio J Med 12: 27-29.
38. Barreiro
R, Regal P (2018) Comparison of the fatty acid profile of Spanish infant
formulas and Galician women breast milk. J Physiol Biochem 74: 127-138.
39. Lv Q,
Sun L, Cui Y, Yang J (2017) Effects of replacement of methionine in diets with
taurine on growth performance and blood index in broilers. Springer 975:
989-1000.
40. Erbersdorfer
HF, Greulich HG (1984) Determinations of taurine in milk and infant formula
diets. Eur J Pediatr 142: 133-134.
41. Kassim
O, Ako-Anai KA, Martin SK (2000) Inhibitory factors in breast milk, maternal
and infant sera against in vitro
growth of Plasmodium falciparum. J
Trop Pediatr 46: 92-96.
42. Dobbs
K, Dent AE (2016) Plasmodium malaria and antimalarial antibodies in the first
year of life. Parasitology 143: 129-138.
43. Pradel
G, Frevert U (2001) Malaria sporozoites actively enter and pass through rat
Kupffer cells prior to hepatocyte invasion. Hepatology 33: 1154-1165.
44. Sancho
J, Gonzalz E (1986) The importance of the Fc receptors for IgA in the
recognition of IgA by mouse liver cells. Immunology 57: 37-42.
45. Al-Kerwi
EA, Al Hashimi AH, Salman AM (2005) Mother’s milk and hydrogen peroxide. Asia
Pac J Clin Nutr 14: 428-431.
46. Hord
NG, Ghannam JS, Garg HK, Berens PD (2011) Nitrate and nitrite content of human,
formula, bovine and soy milks: Implications for dietary nitrite and nitrate
recommendations. Breastfeed Med 6: 393-399.
47. Jädert
C (2014) Diet and inflammation, the role of nitrate and conjugated linoleic
acid. Akademisk afhandeling, Thesis Karolinska Institutet, 49 Prameela Kannan
Kutty Breastfeeding and risk of parasitic infection - A review. Asian Pac J
Trop Biomed 4: 847-858.
48. Rerksuppaphol
S, Rerksuppaphol L (2010) Lactobacillus
acidophilus and Bifidobacterium
bifidum stored at ambient temperature are effective in the treatment of
acute diarrhea. Ann Trop Pediatr 30: 299-304.
49. Newburg
DS, Pickering (1990) Fucosylated oligosaccharides of human milk protect
suckling mice from heat-stabile E. coli.
J Infect Dis 162: 1075-1080.
50. Ruiz-Palacios
GM, Cervantes LE, Ramos P (1998) Role of human milk lactadherin in protection
against symptomatic rotavirus. Lancet 351: 1160-1164.
51. Sultana
R (2015) Comparison of immunoglobulin levels in human milk, cow milk and
formula milk. Kaav Int J Sci Eng Technol, pp: 25-46.
52. Neville
MC (2000) Lactoferrin secretion into milk: Comparison between bovine, murine
and human milk. J Anim Sci 78: 26-35.
53. Hamosh
M (1998) Protective function of proteins and lipids in human milk. Biol Neonate
74: 163-176.
54. Anand
N, Kanwar RK (2015) Effect of lactoferrin protein on red blood cells and
macrophages: Mechanism of parasite-host interaction. Drug Des Devel Ther 9:
3821-3835.
55. Lima
MF, Kierszenbaum F (1985) Lactoferrin effects on phagocytic cell function. I.
Increased uptake and killing of an intracellular parasite by murine macrophages
and human monocytes J Immunol 134: 4176-4183.
56. Shakibei
M, Frevert U (1996) Dual interaction of malaria circumsporozoites protein with
the low density lipoprotein receptor. J Exp Med 184: 1699-1711.
57. Ozturkoglu‐Budak
S (2016) Effect of different treatments on the stability of lysozyme,
lactoferrin and β‐lactoglobulin in donkey's milk. Int J Dairy Technol.
58. Sinnis
P, Thomas EW (1996) Remnant lipoproteins inhibit malaria sporozoite invasion of
hepatocytes. J Exp Med 184: 945-954.
59. Tokumasu
F, Graciela RO (2012) Modifications in erythrocyte membrane zeta potential by Plasmodium falciparum infection. Exp
Parasitol 131: 245-251.
60. Vercellotti
GM, van Asbeck BS, Jacob HS (1985) Oxygen radical-induced erythrocyte hemolysis
by neutrophils: Critical role of iron and lactoferrin. J Clin Invest 76:
956-962.
61. Lee
SB (2016) Antifungal activity of bee venom against Candida albicans. J Pharmacopuncture 19: 45-50.
62. Sritharan
M (2016) Iron homeostasis in Mycobacterium
tuberculosis: Mechanistic insights into siderophore-mediated iron uptake. J
Bacteriol 198: 2399-2409.
63. Posey
JE, Gherardini FC (2000) Lack of a role for iron in the Lyme disease pathogen.
Science 288: 1651-1653.